A serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. A ten-fold serial dilution could be 1 M, 0.1 M, 0.01 M, 0.001 M .. Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in concentration curves with a logarithmic scale. A tenfold dilution for each step is called a logarithmic dilution or log-dilution, a 3.16-fold (100.5-fold) dilution is called a half-logarithmic dilution or half-log dilution, and a 1.78-fold (100.25-fold) dilution is called a quarter-logarithmic dilution or quarter-log dilution. Serial dilutions are widely used in experimental sciences, including biochemistry, pharmacology, microbiology, and physics.
Nov 04, 2017 As another example, a 2-fold dilution is the same as a dilution factor of 2. Therefore, dilution by any factor X is equivalent to X-fold dilution. Please note that the dilution factor calculator below uses the metric unit for volume (fL, pL, nL, μL, mL, and L). Feb 02, 2009 2-fold Dilution Setup? You are given a 50mg/mL solution of bacteria. Explain how you would set up 2 fold dilutions. How would you do the dilutions so that the final volume in the last tube is 500 micro liters? Then, explain if you had a total of 4 tubes, what would be the concentration in. Jun 17, 2014 Solution 2 Remember that serial dilutions are always made by taking a set quantity of the initial dilution and adding it successively to tubes with the same volume. So you multiply each successive dilution by the dilution factor. A serial dilution is a sequence of dilutions created using the same dilution factor. For instance, creating a two-fold dilution with a starting concentration of 10 µM yields the following concentrations: 10 µM, 5 µM, 2.5 µM, 1.25 µM, etc.
In biology and medicine[edit]
2 Fold Dilution Example
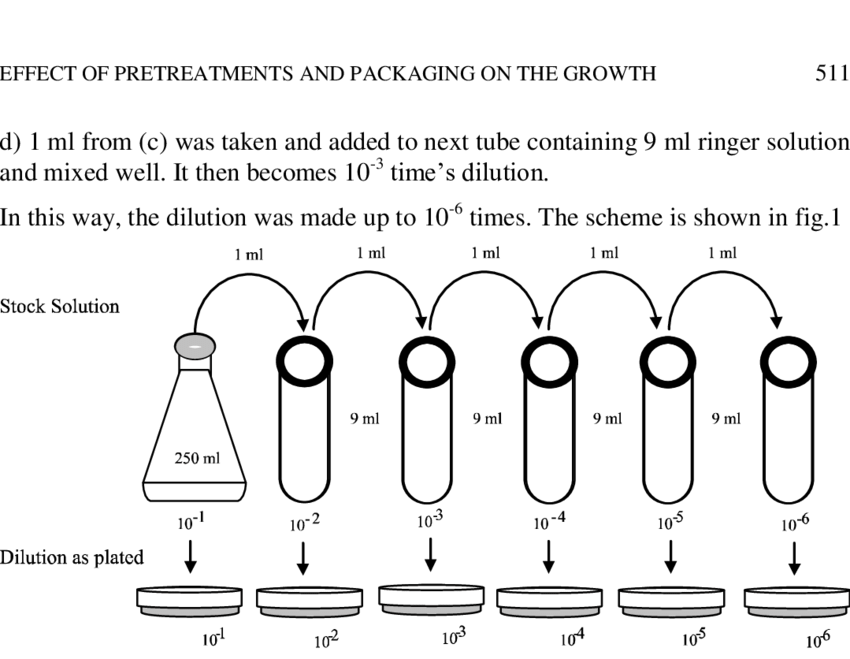
A two-fold dilution reduces the concentration of a solution by a factor of two that is reduces the original concentration by one half. A series of two-fold dilutions is described as two-fold serial dilutions. Dilutions: Explanations and Examples of Common Methods. There are many ways of expressing concentrations and dilution. The following is a brief explanation of some ways of calculating dilutions that are common in biological science and often used at Quansys Biosciences. Using C 1 V 1 = C 2 V 2.
Now you can, with a new way to play the classic game of RISK. Take the battle online with Global Domination mode (must be aged over 13). Defend your territories, draft troops, and launch attacks for the ultimate conquest against friends, family, and foes. Try the game for free. Play Free Risk Online. Description: World domination by strategy propels this all new version of the classic board game. 

I have recently explored the FATE system and have been enjoying the streamlined roleplaying experiences derived from it.Times have changed and I am starting to doubt that any of the D&D versions from advanced to 4e is something an RPGer in today's market wants anymore.Think of it like this- We now have these amazing boardgames with miniatures and pre-balanced systems, all the tactical fun of combat and enough roleplaying. The final playtest version was stripped down too much for my tastes. I assume (hope!) the more interesting bits will be added back in as modular elements.The biggest piece was that they homogenized the classes to essentially be characters with special powers (i liken this to everyone being a mage, though thematically not true, mechanically, it certainly is). Basic dd rules pdf.
In biology and medicine, besides the more conventional uses described above, serial dilution may also be used to reduce the concentration of microscopic organisms or cells in a sample. As, for instance, the number and size of bacterial colonies that grow on an agar plate in a given time is concentration-dependent, and since many other diagnostic techniques involve physically counting the number of micro-organisms or cells on specials printed with grids (for comparing concentrations of two organisms or cell types in the sample) or wells of a given volume (for absolute concentrations), dilution can be useful for getting more manageable results.[1] Serial dilution is also a cheaper and simpler method for preparing cultures from a single cell than optical tweezers and micromanipulators.[2]
In homeopathy[edit]
Serial dilution is one of the core foundational practices of homeopathy, with 'succussion', or shaking, occurring between each dilution. In homeopathy, serial dilutions (called potentisation) are often taken so far that by the time the last dilution is completed, no molecules of the original substance are likely to remain.[3][4]
See also[edit]
References[edit]
- ^K. R. Aneja. Experiments in Microbiology, Plant Pathology and Biotechnology. New Age Publishers, 2005, p. 69. ISBN81-224-1494-X
- ^Booth, C.; et al. (2006). Extremophiles. Methods in microbiology 35. Academic Press. p. 543. ISBN978-0-12-521536-7.
- ^Weissmann, Gerald (2006). 'Homeopathy: Holmes, Hogwarts, and the Prince of Wales'. The FASEB Journal. 20 (11): 1755–1758. doi:10.1096/fj.06-0901ufm. PMID16940145. Retrieved 2008-02-01.
- ^Ernst, Edzard (November 2005). 'Is homeopathy a clinically valuable approach?'. Trends in Pharmacological Sciences. 26 (11): 547–548. CiteSeerX10.1.1.385.5505. doi:10.1016/j.tips.2005.09.003. PMID16165225.
- Michael L. Bishop, Edward P. Fody, Larry E. Schoeff. Clinical Chemistry: Principles, Procedures, Correlations. Lippincott Williams & Wilkins, 2004, p. 24. ISBN0-7817-4611-6.
External links[edit]
- How to Make Simple Solutions and Dilutions, Bates College
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Serial_dilution&oldid=904833671'
